From Lipoic Acid to 1,2-Dithianes: Expanding Radical Ring-Opening to Less-Activated Monomers Such as Vinyl Acetate
Parker T. Morris, Sarah C. Olsen, Marvin Santiago, Hanlin M. Wang, Elliott H. Denton, Javier Read de Alaniz, Christopher M. Bates, Kaitlin R. Albanese, Craig J. Hawker
J. Am. Chem. Soc. March 2026
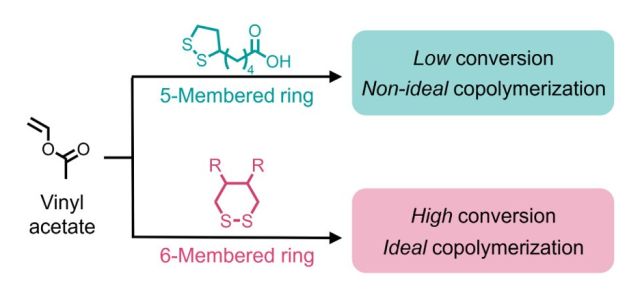
Abstract: Polymers containing cleavable functionality along the backbone represent a path to reduce the environmental persistence of commodity rubbers and plastics. One route to installing cleavable functionality involves the radical ring-opening polymerization of five-membered 1,2-dithiolanes such as α-lipoic acid, which introduces disulfide bonds into vinyl polymer backbones. However, the ring strain and electronics of 1,2-dithiolanes generally restrict reactivity to only favor copolymerization with more-activated comonomers, such as acrylate and styrene derivatives. Here, we show that the six-membered cyclic disulfide, 1,2-dithiane overcomes this limitation under simple thermal free-radical conditions. We demonstrate that 1,2-dithiane copolymerizes efficiently with the less-activated monomer vinyl acetate. In sharp contrast to α-lipoic acid and its derivatives, 1,2-dithiane exhibits nearly ideal copolymerization across different feed ratios, affording high conversions of both monomers (>90%), tunable molar masses (Mn ≈ 10–100 kg mol–1), and scalability (>20 g). Moreover, the 1,2-dithiane scaffold is synthetically versatile: a 1,2-dithiane-4,5-diol was synthesized from dithiothreitol as a building block for further derivatization. By varying dithiane functionality and loading, poly(dithiane-co-vinyl acetate) copolymers span semicrystalline to elastomeric mechanical properties and, critically, embed backbone-cleavable sulfur motifs (including monothioacetal units) even at low (<1 mol %) loadings. This operationally simple reaction highlights the key influence of ring size in the copolymerization behavior of disulfide-containing monomers and demonstrates the practical advantages of using 6-membered cyclic disulfides as renewable building blocks for creating degradable vinyl copolymers derived from inexpensive, industrially relevant feedstocks.